Phylogenetic analysis of pufferfish Rh proteins and tissue distribution... | Download Scientific Diagram

Acid-sensitive ionic channels in midbrain dopamine neurons are sensitive to ammonium, which may contribute to hyperammonemia damage | PNAS

SOLVED: What is the most efficient way to increase the pH and buffer capacity of an ammonium (NH4+) /ammonia (NH3) buffer? dilute the buffer add ammonium chloride add ammonia add NaOH(aq) add

Acid-sensitive ionic channels in midbrain dopamine neurons are sensitive to ammonium, which may contribute to hyperammonemia damage | PNAS

Acid-sensitive ionic channels in midbrain dopamine neurons are sensitive to ammonium, which may contribute to hyperammonemia damage | PNAS

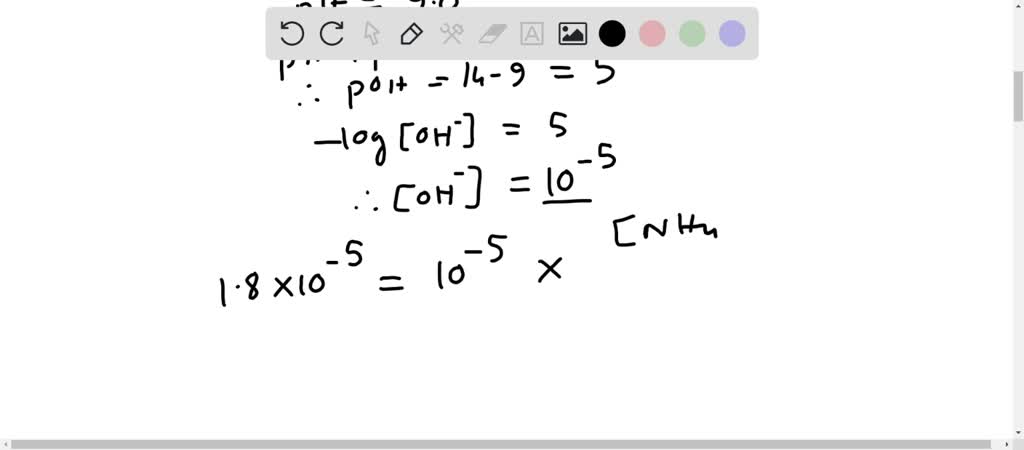
Calculate the pH of a buffer prepared by mixing 300 cc of 0.3 M NH3 and 500 cc of 0.5 M NH4Cl . Kb for NH3 = 1.8 × 10^-5